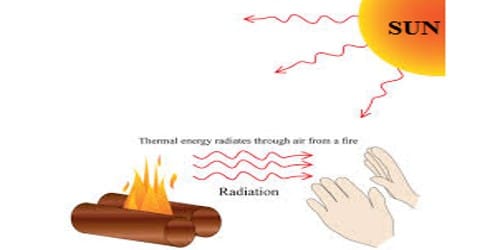
Prevost applied the idea of thermal equilibrium to radiation. According to him, the rate at which a body radiates or absorbs heat depends on the nature of its surface, its temperature and the temperature of the surroundings. The total amount of heat radiated by a body increases as its temperature rises. He suggested that all bodies radiate energy but hot bodies radiate more heat than the cooler bodies. A body at a higher temperature radiates more heat energy to the surroundings than it receives from the surroundings. That is why we feel warm when we stand before the furnace. For example, if you touch someone, they might feel your skin as either hot or cold.
A body at high temperature radiates more heat to the surroundings than it receives from it. Similarly, a body at a lower temperature receives more heat energy than it loses to the surroundings. That is why we feel cold when we stand before an ice block. In this case, the amount of heat absorbed by the body from the enclosure per second is greater than that emitted by the body at the same time so that there is a net gain of heat by the body. At one point in time, the rate of exchange of heat from both bodies will become the same. Now the bodies are said to be in ‘thermal equilibrium’.
Thus the rise or fall of temperature is due to the exchange of heat radiation. When the temperature of the body is the same as that of surroundings, the exchanges of heat do not stop. In such a case, the amount of heat energy radiated by the body is equal to the amount of heat energy absorbed by it. A body at a higher temperature than the surroundings radiates heat at a faster rate than it absorbs. The rate of emission of heat by a body depends upon its absolute temperature.
Only at absolute zero temperature, a body will stop emitting. A body will stop emitting radiation only when it is at absolute zero. Therefore Prevost theory states that all bodies emit thermal radiation at all temperatures above absolute zero irrespective of the nature of the surroundings. Example: 0 K or -273° C. At this temperature the kinetic energy of the molecule is zero. The rate of emission of heat by a body does not depend upon the temperature of its surroundings. So, It loses heat and its temperature falls. And it gains heat, its temperature rises. A body that is at a lower temperature than the surroundings absorbs heat at a faster rate than it radiates. Hence, there is no net loss or gain of heat. Its temperature is unchanged.
Therefore, Prevost’s theory states that all bodies emit thermal radiation at all temperatures above absolute zero, irrespective of the nature of the surroundings. In the case of thermal equilibrium, the process of radiation and absorption continues to take place. Every material body, at any temperature above absolute zero, radiates heat to the surroundings and at the same time absorbs heat from the surroundings.