The basic conception of the molecular kinetic theory of matter is to consider that the molecules of a matter are in motion. The kinetic theory of matter is based on the following postulates:
- Any substance consists of innumerable number of minute particles. These particles are called molecules.
- The molecules are so small that they are considered as points.
- The molecules of a substance are always in motion.
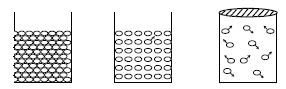
- The gaseous molecules remain at a considerable distance. For this reason almost no force of attraction and repulsion act between them. Though the liquid molecules remain at some distance, force of attraction between them prevails. This cause compels the liquid to take the shape of the container in which it is kept. In solid substances the particles remain very close to each other and very strong attractive force exists in them which give the solid substance a definite size and volume.
- The molecules move at random in liquid and gas.
So, these molecules collide among themselves and also on the surface of the walls of the container.