Raman Spectrum: The Raman effect can be easily understood, by considering the scattering of the photon of the incident light with the atoms or molecules.
Let the incident light consist of photons of energy hν0.
- If a photon strikes an atom or a molecule in a liquid, part of the energy of the incident photon may be used to excite the atom of the liquid and the rest is scattered. The spectral line will have the lower frequency and it is called stokes line.
- If a photon strikes an atom or a molecule in a liquid, which is in an excited state, the scattered photon gains energy. The spectral line will have the higher frequency and it is called Anti−stoke’s line.
- In some cases, when a light photon strikes atoms or molecules, photons may be scattered elastically. Then the photons neither gain nor lose energy. The spectral line will have the unmodified frequency.
If ν0 is the frequency of incident radiation and νs the frequency of scattered radiation of a given molecular sample, then Raman Shift or Raman frequency Δν is given by the relation Δν = ν0 – νs.
The Raman shift does not depend upon the frequency of the incident light but it is the characteristic of the substance producing Raman effect. For Stoke’s lines, Δν is positive and for Anti–stoke’s lines Δν is negative. The intensity of Stoke’s line is always greater than the corresponding Anti−stoke’s Line. The different processes giving rise to Rayleigh, Stoke’s and Anti-stokes lines are shown in Figure.
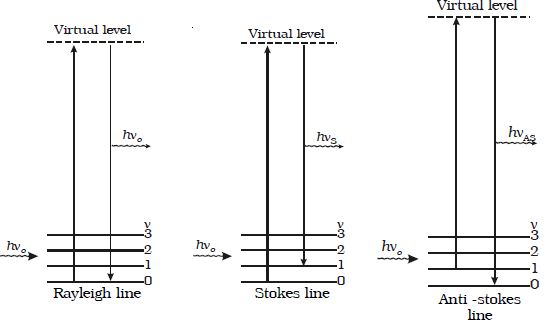
(ν = 0, 1, 2 …. are the vibration levels of the ground electronic state.)
Fig: Raman Spectrum
When a system interacts with a radiation of frequency νo, it may make an upward transition to a virtual state. A virtual state is not one of the stationary states of the molecule. Most of the molecules of the system return back to the original state from the virtual state which corresponds to Rayleigh scattering. A small fraction may return to states of higher and lower energy giving rise to Stoke’s line and Antistoke’s line respectively