A triatomic Molecule non-linear molecule may rotate, about the three mutually perpendicular axes, as shown in Figure.
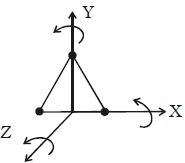
Therefore, it possesses three degrees of freedom of rotation in addition to three degrees of freedom of translation along the three co-ordinate axes. Hence it has six degrees of freedom.
Examples: molecules of H2O, SO2 etc.
In all the Triatomic Molecule (Non-linear type) cases, only the translatory and rotatory motion of the molecules has been considered. The vibratory motion of the molecules has not been taken into consideration.