Carrot’s cycle is reversible
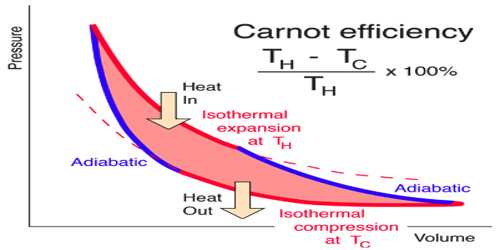
The cycle in which an ideal gas used as a working substance starts at constant volume, pressure, and temperature passes through an isothermal and adiabatic expansion and then through an isothermal and adiabatic compression returns to the initial state is called a Carnot’s cycle.
The Carnot cycle is not the only possible reversible process. It is famous because historically, the idea of the Carnot cycle crystallized the concept of reversible vs. irreversible processes. It is probably also the simplest example of how to reversibly transform heat to work or vice versa.
In practice, reversible cycles are unrealistic because each process is associated with irreversibility, for example, friction. However, the upper limit on the performance of real cycles can be obtained by using the corresponding reversible cycles. And the most well-known reversible cycle process is the Carnot cycle.
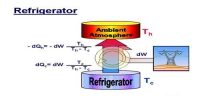
The Carnot heat-engine cycle described is a totally reversible cycle. That is all the processes that comprise it can be reversed, in which case it becomes the Carnot refrigeration cycle.
Characteristics, which are needed be present in a cycle to become reversible, are present in an ideal Carnot’s engine, for example-
- There is no friction between the piston and the cylinder,
- Processes applied to the working substance (gas) are executed very slowly.
- In making the piston and the cylinder ideal heat insulator and ideal heat conductor are used and the heat source and heat sink are made of materials having the very high thermal capacity so that the isothermal processes occur at a constant temperature.