If the free surface of a liquid is plane, then the surface tension acts horizontally (Fig. a). It has no component perpendicular to the horizontal surface. As a result, there is no pressure difference between the liquid side and the vapour side.
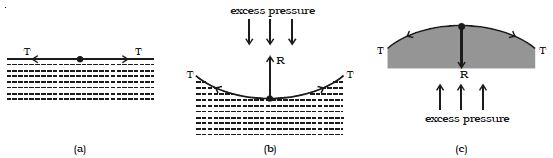
Fig: Excess of pressure across a liquid surface
If the surface of the liquid is concave (Fig. b) then the resultant force R due to surface tension on a molecule on the surface act vertically upwards. To balance this, an excess of pressure acting downward on the concave side is necessary. On the other hand if the surface is convex (Fig. c), the resultant R acts downward and there must be an excess of pressure on the concave side acting in the upward direction.
Thus, there is always an excess of pressure on the concave side of a curved liquid surface over the pressure on its convex side due to surface tension.