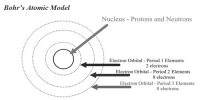
Pauli’s exclusion principle states that “No two electrons in an atom can have the same values for the 4 quantum numbers. That is in an atom any two electrons can have 3rd quantum number same but their 4th quantum number must be different from each other.”
Their size of energy levels, shapes of energy levels and three dimensional orientations would be same but spins must be different.
Example:
He (2)→ 1s2
For 1st electron, n = 1, l = 0 m = 0, S = +1/2
For 2nd electron, n = 1, l = 0 m = 0, S = -1/2
i.e. An orbital can hold only two electrons of opposite spin this leads to the conclusion that an orbital can have maximum of two electrons. Thus Pauli’s exclusion principle also states that an orbital can have maximum two electrons of opposite spin.