This is a phenomenon in which certain organic or inorganic compounds have the property of rotating plane polarised light. The compounds which exhibit this property are called optical isomers.
The optical isomers of a compound have identical physical and chemical properties. The only distinguishing property is that the isomers rotate the plane of polarised light either to the left or right. In a coordination compound of type [PtCl2(en)2]12±, two geometrical isomers are possible. They are cis and trans. Among these two isomers, cis isomer shows optical activity because the whole molecule is asymmetric.
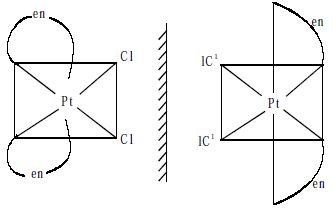
Fig: Optical isomers of cis (PtCl2(en)2]2+