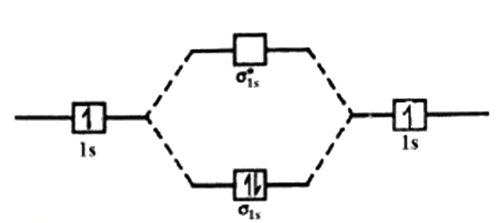
Hydrogen molecule (H2) is formed by the combination of two hydrogen atoms. Each hydrogen atom in the ground state has one electron in 1s orbital. Therefore, in all there are two electrons in hydrogen molecule which are present in lower most σ1s molecular orbital. According to Pauli’s exclusion principle, these two electrons should have opposite spins.
The molecular orbital electronic configuration of hydrogen molecule is (σ1s)2.
The molecular orbital energy level diagram of H2 molecule is given in Figure.