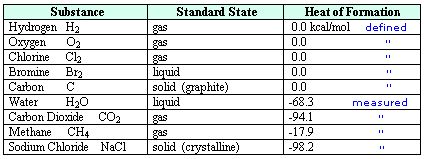
In order that chemists worldwide can compare notes on thermochemical experiments, a series of special conditions have been agreed. The term “standard state enthalpy” refers to an enthalpy change for a reaction in which reactants and products are considered to be in their standard states (most stable state of the substance) at a specified temperature of 298 K and pressure of 1 atmosphere. The standard enthalpy of formation refers to the enthalpy change when one mole of a compound is formed from its elements.
Standard State Enthalpies: Enthalpy change is the name given to the amount of heat evolved or absorbed in a reaction carried out at constant pressure. It is given the symbol ΔH, read as “delta H”. The enthalpy change for a reaction in which reactants are in their standard states is denoted as; AH° (“delta H zero” or “delta H nought”).
Measuring the Enthalpy changes in chemical reactions: Enthalpy changes are measured by measuring the heat required to raise the temperature of a surrounding substance (usually water), because a thermochemical measurement is based on the relationship between heat and temperature change. The heat required to raise the temperature of a substance is its heat capacity. The standard enthalpy of reaction occurs in a system when one mole of matter is transformed by a chemical reaction.














