The Nature of Thermodynamics
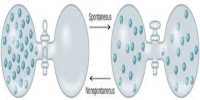
Thermodynamics is an important discipline in physical science. It deals with the relationship between thermal energy and other forms of energy. The science of thermodynamics was developed in the nineteenth century and is based on innumerable observations and experiments over many years. The deductions from these observations and experiments are summarized in the form of three fundamental laws: the first, the second and the third law of thermodynamics. The laws of thermodynamics are accepted as laws because no exception to these has ever been found by experiments. There are two approaches to the study of thermodynamics:
Classical thermodynamics- deals with the macroscopic views of the matter, i.e., the properties of matter in bulk. It does neither consider the internal constitution of matter nor does it require any theory of matter.
Statistical thermodynamics- deals with the study of the macroscopic behaviors of thermodynamic systems using probability theory. Statistical thermodynamics provides a molecular level interpretation of thermodynamic quantities such as work, heat, energy and entropy. The development of this branch of thermodynamics was initiated by an Austrian physicist Ludwig Boltxmann in 1870.