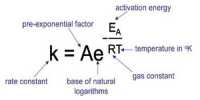
Question 1:
Consider an N2 molecule at 298 K moving with a velocity of 515 m s-1. In the absence of any collisions, how long will it take the molecule to travel 10 km? This absence of collisions corresponds to an extremely low pressure.
Velocity = (distance/time); so, V = d/t
Question 2:
Remember that acceleration is the differential of the velocity-time curve (v-t graph) at a particular point in time.
Acceleration = [d(Velocity) / d(time)]; so, a = dv/dt
Note that the symbol for differential is a roman (upright) “d” as it is not a variable but rather indicates an operation to be carried out on the variable, it is an “operator”. The N2 molecule of gas collides with another molecule in a collision lasting 100 picoseconds (symbol ps and 1 ps =10-12 s). As a result of the collision, the velocity of our N2 molecule increases from 515 to 630 m s-1. What is the acceleration of the molecule if it is still travelling in the same direction?
Solution 1:
V = d/t; t = d/V = (10000 m /515 ms-1)
So, t = 19.4 s
Notice how in the absence of any collision a molecules can travel a long distance very quickly, e.g. inside a mass spectrometer flight-tube where the pressure may be 10-9 atmospheres or less, or in space. Of course, at any ambient pressures the molecules are rapidly colliding with one another and changing their directions as well as their speeds but nevertheless the average speed of N2 at room temperature is still around 515 m s-1.
Solution 2:
Acceleration, a, is the gradient of the velocity-time graph (v-t graph) which in this case is
a = dv/dt; a = [630 m s-1 – 515 m s-1] / 100x 10-12 s
so, a = 1.15 x 1012 m s -1
Notice how molecules of gas are capable of very rapid acceleration (also deceleration) by means of collisions.