Langmuir Isotherm Measurement
Many systems covering a wide range of temperature obey the Langmuir isotherm. As a matter of fact, deviation from linearity was ascribed to various other reasons which were not considered in the Langmuir theory.
The theory: “Whenever a gas is in contact with a solid there will be an equilibrium established between the molecules in the gas phase and the corresponding adsorbed species (molecules or atoms) which are bound to the surface of the solid.”
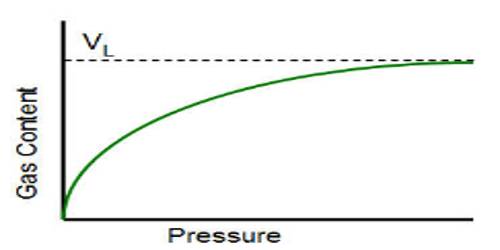
One typical Langmuir isotherm of the adsorption of sulphur dioxide on barium chromate is shown in Figure. Strict obedience to Langmuir theory can be seen along the straight line ‘ab’. After the point, ‘b’ is reached deviation occurs Langmuir isotherm is extensively used for the measurement of surface area or particle size of the powdered material. From the slope and intercept of Langmuir plot, the volume of the gas required to form a unimolecular layer on the surface of the powder is calculated. Since the area of the molecules of the absorbed gas is known the area of the surface of the solid can be easily calculated.
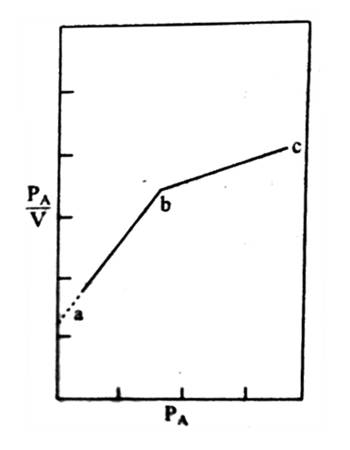
Figure: Langmuir plot
The results are expressed in terms of surface area in cm2/g or m2/g of the adsorbent. Surface area of such materials as paper, pulp, rayon, cotton, jute etc., can be determined by use of Langmuir equation as the method is equally applicable in case of adsorption from solution.