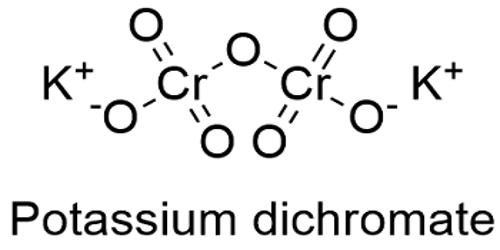
Potassium dichromate is an important chemical used in industries as an oxidizing agent and for the preparation of many other compounds. It occurs naturally in mineral form as lopezite, a very rare mineral. It is used in many applications as an oxidizing agent and is also used in the preparation of different products such as waxes, paints, glues, etc.
Preparation of potassium dichromate from chrome iron ore.
Dichromates are usually prepared from chromates and this is obtained by the combination of chromite ore with sodium/potassium carbonate in the presence of air. The preparation of Potassium dichromate (K2Cr2O7) from its ore involves the following steps:
(i) Conversion of chrome iron ore to sodium chromate
The powdered ore is mixed with Na2CO3 and quick lime, then roasted in a reverberatory furnace with free expose to air.
4FeO.Cr2O3 + 8Na2CO3 + 7O2 → 8Na2CrO4 + 2Fe2O3 + 8CO2↑
(ii) Conversion of Na2CrO4 to Na2Cr2O7
Sodium chromate solution so obtained is filtered and treated with con.H2SO4, when sodium chromate is converted to sodium dichromate.
2Na2CrO4 + H2SO4 → Na2Cr2O7 + Na2SO4 + H2O
(iii) Conversion of sodium dichromate into potassium dichromate
Potassium dichromate is produced industrially by reacting potassium chloride (KCl) with sodium dichromate (Na2Cr2O7). It is also obtained from its related compound, potassium chromate (K2CrO4), which reacts with acids to give the dichromate salt. A hot concentrated solution of sodium dichromate is treated with KCl, when potassium dichromate, being much less soluble than sodium salt, crystallizes out on cooling.
Na2Cr2O7 + 2KCl → K2Cr2O7 + 2NaCl
Physical properties: It is a bright red-orange crystalline solid with a density of 2.676 g/mL, the melting point of 398 °C and boiling point of 500 °C, when it decomposes. It is odorless and highly water-soluble.
Uses: Potassium dichromate is used for preparing strong cleaning solutions for glassware and for etching materials. It also has uses in leather tanning, photographic processing, cement, and wood staining. It is used as an oxidizing agent in many applications and is also used to prepare various products such as waxes, paints, glues, etc.