Isothermal and adiabatic processes
Isothermal and adiabatic processes are common terms in thermodynamic while discussing the energy variation in form of heat.
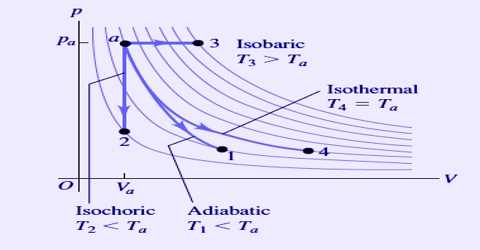
A process in which the temperature is maintained constant during the change is called an isothermal process. In an isothermal process dT = 0. In such a case heat is allowed to flow from the surrounding into the system during expansion of the gas, and taken out from the system to the surrounding during compression of the gas. An isothermal process is usually carried out by placing the system in a thermostat (constant temperature bath). There may be energy flow into and out of the system, however only the amount required to keep the temperature of the system constant. For Example: Phase changes – melting solids and boiling liquids of pure substances requires substantially energy transfer, but does not change temperature.
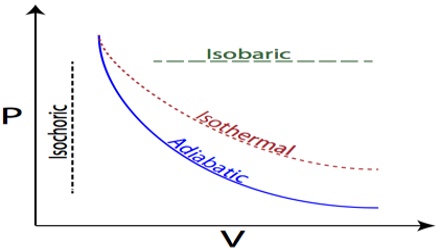
On the other hand, in an adiabatic process heat is not allowed into or out of the system during a change. The system is completely isolated from the surroundings by an insulated boundary. In an adiabatic process dq = 0.
For example: Analysing the stroke of a piston where heat transfer outside of the system can be minimal due to the short period of time analyzed.