Ideal Solution
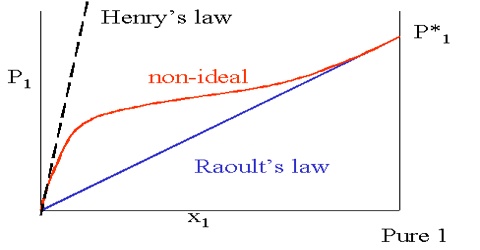
As we know, solution is said to be ideal when it obeys Raoult’s law over all compositions and at all temperatures. Such a solution is formed from its components without heat being given out or absorbed (∆H=0) and they solution process is not accompanied by any volume change (∆V=0). It follows from Raoult’s law that if the solution is ideal the escaping tendency of each component will be proportional to the mole fraction of that component in the solution.

From the molecular point of view one can say that in an ideal solution of A and B the intermolecular forces between A and A molecules, A and B molecules, and B and B molecules are the same and the escaping tendency of the A molecules will be the same whether these are surrounded by other A molecules or B molecules or partly by A and partly by B molecules. If there is interaction the solution will deviate from ideal behavior. In reality there are very few solutions which obey Raoult’s law strictly, i.e., behave ideally; but the more dilute a solution is the more nearly dues it approach ideality. When the solution is infinitely dilute it is expected to be ideal.