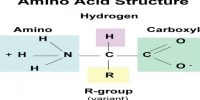
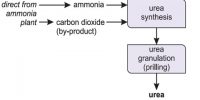
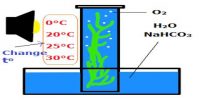
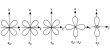
Chemical equilibrium: Chemical equilibrium is the state where the rate of forward and backward reaction will be equal in the reversible reaction. Equilibrium is when the rate of the forward reaction equals the rate of the reverse reaction.
The laws of chemical equilibrium define the direction in which a chemical reaction will proceed, as well as the quantities of reactants and products that will remain after the reaction comes to an end.
- Chemical equilibrium occurs at only reversible reaction.
- The reaction is not stopped at equilibrium but it actually runs both sides equally.