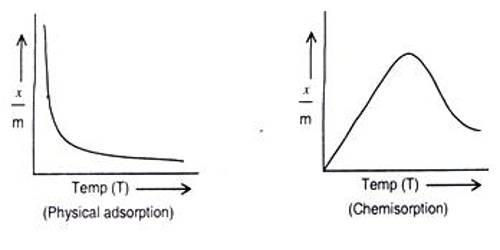
Difference between Physical adsorption and Chemisorption
Physical adsorption is a process in which the electronic structure of the atom or molecule is barely perturbed upon adsorption. Chemisorption or chemical adsorption is adsorption in which the forces involved are valence forces of the same kind as those operating in the formation of chemical compounds
Physical Adsorption
- Adsorption is rapid and equilibrium is quickly established.
- Adsorption usually takes place quickly at low temperature.
- Enthalpy of adsorption is low, usually 0 – 40 kJ mol-1.
- Adsorption is exothermic; energy is released from the system due to loss of kinetic energy.
- The temperature coefficient is negative, i.e., amount of adsorption decreases due to rising in temperature.
- Little or no activation energy is involved.
- The process is nearly reversal. On removing the surface the molecular species can be recovered.
- Adsorption is not specific. Any solid surface can adsorb any gaseous substance. Adsorption is universal.
- Physical adsorption is not associated with catalysis.

Chemisorption
- Adsorption is rather slow and equilibrium is slowly established.
- Adsorption usually takes place at low temperature.
- Enthalpy of adsorption is large, comparable to the enthalpy of reaction.
- Energy release is not only due to the loss of kinetic energy but also due to chemical interactions.
- The temperature coefficient is positive, i.e., amount of adsorption increases due to loss of temperature.
- Activation energy is large.
- The process is, in most cases, irreversible. The same molecular species cannot be completely recovered, they contain some reaction products.
- Adsorption is specific and not universal in character.
- Chemisorption is associated with catalysis.