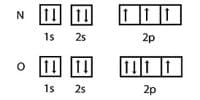
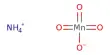Properties of Chromium (Cr):
Physical Properties:
- The metal is silvery white and crystalline.
- It is very hard and brittle
- It melts at 2113K.
Chemical Properties:
Action of air: It is unaffected by air at ordinary temperatures.
When heated to very high temperature at about 2000°C it is oxidized to chromic oxide.
- 4Cr + 3O2 → 2Cr2O3
Action of Water: There is no action at ordinary temperatures. However it decomposes steam at red heat to give chromic oxide and hydrogen.
- 2Cr + 3H2O → Cr2O3 + 3H2
Action of Acids: It dissolves in dilute hydrochloric acid and sulphuric acid to liberate hydrogen and forms chromous salts.
- Cr + 2HCl → CrCl2 + H2↑
- Cr + H2SO4 → CrSO4 + H2↑
With hot concentrated sulphuric acid it gives chromic sulphate and liberates sulphur dioxide.
- 2Cr + 6H2SO4 → Cr2(SO4)3 + 3SO2 + 6H2O
Dilute nitric acid does not attack the pure metal while concentrated acid renders it inactive or passive i.e., it does not show its usual reactions.
Action with Halogens: Chromium combines directly with fluorine and dry chlorine to give chromium (III) halides.
- 2Cr + 3F2 → 2CrF3
- 2Cr + 3Cl2→ 2CrCl3