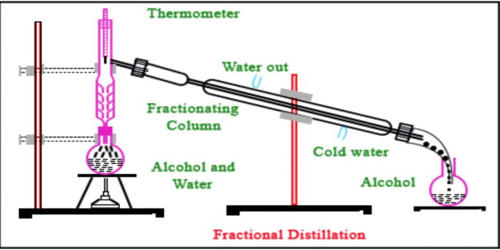
Fractional distillation is the process of taking a chemical mixture and using heat to separate out the various components in that mixture. The principle of fractional distillation is that different liquids boil at different temperatures. It is a process by which components in a chemical mixture are separated into different parts (called fractions) according to their different boiling points. The miscible liquids boil at different temperatures and evaporate at different temperatures.
Principle of Fractional distillation
- Normally the composition of vapor of any liquid mixture does not remain equal to the composition in a liquid state. When the mixture is heated, the liquid with lower boiling point boils and turns into vapors.
- The more volatile component remains more in vapor state than that of the liquid state. The process involves repeated distillations and condensations and the mixture is usually separated into component parts.
- After heating, the more volatile component increase in a vapour state, and when this vapor is liquefied then a more volatile component increase in the liquid state.
- First vaporization and then condensation (liquefaction) are combinedly known as distillation. When this distillation process repeatedly occurs, a time will come when a more volatile component remains in the liquid state at a pure state. In this way components of the liquid-liquid mixture can be separated as a pure substance by using fractional distillation method.
The basic principle of this type of distillation is that different liquids boil and evaporate at different temperatures. So when the mixture is heated, the substance with a lower boiling point starts to boil first and convert into vapors. Let consider a liquid-liquid mixture of A and B where A is more volatile than B this mixture can be separated by the following process:

When you think of this process, the first word that should come to mind is separation. The separation happens when the mixture is heated at a certain temperature where fractions of the mixture start to vaporize. Crude oil is separated into numerous chemicals, such as gasoline, diesel fuel and acetone are examples of fractional distillation. Gasoline and many other chemicals are produced from crude oil using fractional distillation. Crude oil is heated until it evaporates.
Applications
- Fractional distillation is used to purify chemicals and also to separate mixtures to obtain their components.
- It is used for the purification of water as well as separating acetone and water.
- It is used in several industries like oil refineries and chemical plants mainly for purification and separation of many organic compounds.
- It is used in the production of high-purity silicon from chlorosilanes. Silicon is widely used in semiconductors.
- It is used to separate a mixture of two or more miscible liquids that boil without decomposition and for which the difference in boiling points is less than 30℃.