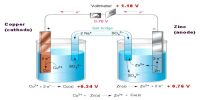
Potential difference is the difference in electric potential (also referred to as electrical pressure) between two points. This difference in electrical potential can be measured by a voltmeter. The volt (symbol V) is the SI unit of potential difference.
The movement of electrons in electro-chemical cells can be compared to water flowing or being pumped from one point to another. Water moves from a point of high pressure to a point of lower pressure. Thus, a pressure difference is required. The work done in moving the water through a pipe depends on the volume of water and the pressure difference.
Electric Potential
Before an electro-chemical cell is connected, a redox reaction is ready to take place but cannot yet proceed. At the anode, the oxidation half-reaction is ready to produce surplus electrons, so there is a high electric potential. At the cathode, the reduction half- equation is ready to use up surplus electrons, so there is a low electric potential.
Redox couples: Each redox half reaction must always refer to a pair of species; the reduced form and the oxidised form.
The reduction potential of the redox couple is a measure of how easily the oxidised species accepts an electron to change to the reduced species.
The oxidation potential is a measure of how easily the reduced form donates an electron to change to the oxidised form. It is the negative of the reduction potential for the reverse reaction.
For example, the reaction Zn → Zn2+ + 2e– happen easily.
Whereas, the reaction Zn2+ + 2e– → Zn does not happen easily.
Therefore, the following redox equilibrium lies well to the left as follows:
Zn2+ + 2e– → Zn
Potential Difference: The difference in electric potential between the two half-cells gives the overall cell voltage. If the two half-cells are connected by a wire and a voltmeter, the potential difference can be measured.