The magnetic properties of a D-Block Elements are determined by the number of unpaired electrons in it. There are various substances which show magnetic activities. Generally, there are two main types of substances. We have substances that are attracted by the magnetic field and are called paramagnetic. This occurrence is called paramagnetism. On the other hand, we also have substances which are repelled by a magnetic field and are called diamagnetic substances. A substance shows diamagnetism when it contains only paired electrons.
(i) Paramagnetic substances
The paramagnetic character arises because of the presence of unpaired electrons. Paramagnetic substances are the substances which are attracted by the magnetic field.
These are softly concerned to a magnetic field. Paramagnetic property is only revealed when the substance contains one or more unpaired electrons. Materials that are concerned by an outwardly applied magnetic field and form internal induced magnetic fields in the path of the applied magnetic field. The paramagnetic character emerges in view of the propinquity of unpaired electrons. Paramagnetic substances are the substances which are pulled in by the magnetic field. When a substance acquires an eternal magnetic moment, it is known as ferromagnetic and the occurrence is called ferromagnetism.
Reason: It presence of unpaired electrons in the atom.
(ii) Diamagnetic Substances
Diamagnetic character arises because of the absence of unpaired electrons. The diamagnetic substance is the substances which are repelled by the magnetic field.
These are repelled to a magnetic field. Materials that generate an induced magnetic field in a path contrary to an outwardly applied magnetic field and are consequently repelled by the applied magnetic field. Diamagnetic character emerges as a consequence of the nonappearance of unpaired electrons. Diamagnetic substances are the substances which are disgusted by the magnetic field.
Reason: It absence of unpaired electrons in the atom.
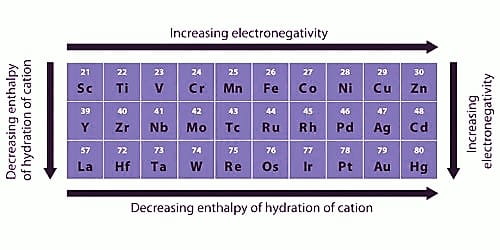
Most of the transition elements and their compounds are paramagnetic and are attracted by the magnetic field. Greater the number of unpaired electrons in the substance greater is the paramagnetic character, The magnetic character of a substance is expressed in terms of magnetic moments. The magnetic moment can be calculated using the relation
μ = √{n(n + 2)} BM (BohrMagneton)
n = number of unpaired electrons
Example
Ti3+ – The number of unpaired electrons is 1. Hence
μ = √{1(1+ 2)} BM = 3 = 1.732 B.M
Larger the value of the magnetic moment, the greater is the paramagnetic character.
In addition to paramagnetic and diamagnetic substance, there are a few substances such as iron which are highly magnetic as compared to ordinary metals.
These substances are called ferromagnetic substances.