Formation of coloured ions of of d-block elements:
An electron jumps from one d-orbital to another. Most of the transition metal compounds are colored in their solid or solution form. The color of transition metal ions is due to the presence of unpaired electrons in it and the energy gap between two energy levels in the same d-subshell being small. When an electron from a lower energy d orbital is excited to a higher energy d orbital, the energy of excitation corresponds to the frequency of light absorbed. This frequency generally lies in the visible region.
In complexes of the transition metals, the d orbitals do not all have the same energy. Hence a very small amount of energy is required for excitation of electrons from one energy level to the other. The energy can be easily provided by the visible light. The color observed corresponds to the complementary color of the light absorbed. The pattern of the splitting of the d orbitals can be calculated using crystal field theory.
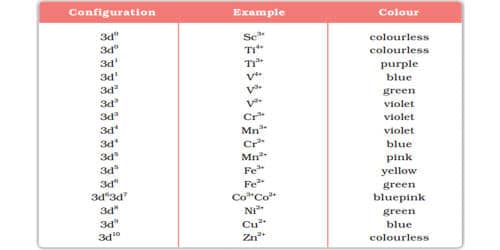
The color of some transition metal ions are given in the Table.
Formation of Coloured Ions –
- An electron from a lower energy d orbital is excited to a higher energy d orbital, the energy of excitation corresponds to the frequency of light absorbed.
- This frequency generally lies in the visible
- The color of the transition metal ions is due to the presence of unpaired or incomplete d-orbitals.
- The absorption of visible light and hence the colored nature of the transition metal cations is due to the promotion of one or more unpaired d-electron from a lower to a higher level within the same d-subshell. This promotion requires a small amount of energy available in the visible light.
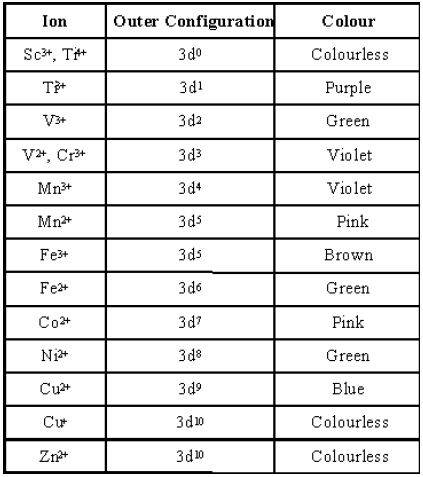
- Sc3+, Ti4+, Cu+, and Zn2+ have either entirely empty or entirely filled 3d-orbital, i.e. they do not have any unpaired d-electron, and hence appear colorless.
It may be noted that Zinc, Cadmium, and Mercury salts do not form any colored compounds because of the absence of vacant d orbitals to which electrons can be excited. Sc3+ ions are also colorless because of the absence of d-electrons. The color observed corresponds to the complementary color of the light absorbed.