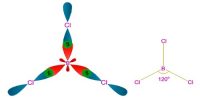
Valence bond theory, primarily the work of Linus Pauling regarded bonding as characterized by the overlap of atomic or hybrid orbitals of individual atoms.
Although VB theory was the principal way in which chemist visualized coordination compounds until the 1950s, it has fallen into disfavor due to its inability to account for various magnetic, electronic, and spectroscopic properties of these compounds.
Octahedral (d2sp3 or sp3d2), tetrahedral (sp3) and square planar (dsp2) complexes of d1, d2, d3, d9 ions have the same number of unpaired electrons and hence cannot be distinguished from each other on the basis of the number of unpaired electrons. Similarly, outer orbital tetrahedral and octahedral complexes of all ions namely d1, d9 which have the same number of unpaired electrons cannot be distinguished from each other.
Valence Bond Theory does not explain the behavior of complexes having d8 central ion (Ni+2, Pd+2, and Au+3) in forming the unexpected five coordinated complexes. Moreover, it is not clear why this theory prefers only square planar geometry such as tetrahedral
Defects of Valence bond theory
- The main defect of the simple Valence bond theory lies in its failure
- To include the antibonding molecular orbitals produced during complex formation.
- Failure to explain the tetravalency exhibited by carbon
- No insight offered on the energies of the electrons.
- The theory assumes that electrons are localized in specific areas.
- It does not give a quantitative interpretation of the thermodynamic or kinetic stabilities of coordination compounds.
- No distinction between weak and strong ligands.
- No explanation for the color exhibited by coordination compounds.
- It fails to offer an explanation for the striking colors of many complexes, which arise from their selective absorption of light of only certain wavelengths
Another common cause in which VB theory will struggle is in the prediction of bonding in transition metal compounds as these undergo different types of bonding from most non-metals. VSEPR also fails when looking at transition metals due to the change from VB theory to MO theory.