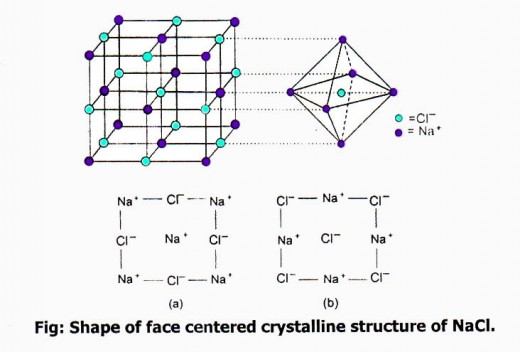
Lattice Structure of NaCl:
In the lattice structure of NaCl, Na+ is surrounded by six oppositely charged Cl– and each Cl– is surrounded by six Na+ forming an octahedron. At the centre of octahedron, there is one ion and at the corners there are six opposite ions. By the internal 3D arrangement of such innumerable octahedrons the giant 3D cubic lattice of NaCI is formed.
By the X-ray analysis it is seen that Na+ and Cl– are arranged in two cubic lattice structure. The upper and lower faces of one cube is as fig. a and the upper and lower feces of another cube is as fig. b. One cube is interpenetrated into another cube. As a result the cube unit cell of NaCI lattice structure as shown in fig is formed.
The unit cell is also formed by eight small cubes and at the corners of these unit cells there are eight ions. as a whole by the innumerable number of cubic unit cells the cubic lattice structure of NaCI is formed. NaCI never exists as a single molecule, in NaCI structure the co-ordination number of Na+ or Cl– is six.