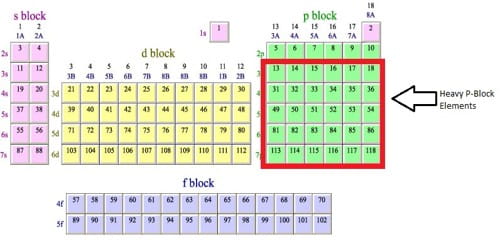
The p-block is on the right side of the periodic table and includes elements from the six columns beginning with column 3A and ending with column 8A. Helium, which is at the top of column 8A, is not included in the p-block. In the periodic table shown here, the p-block is colored orange. The atoms of elements of these groups receive their last electron in 2p, 3p, 4p, 5p,6p, and 7p orbitals.
General characteristics of p-block elements
- The general electronic configuration of p-block elements is ns2 np1-6. The p-block elements have relatively little in common except that their outer electrons are in p orbitals.
- These elements include metals and non-metals with a few semimetals (Metalloids). This contrasts with the considerable similarities in properties of the elements of the s-block and also between those of the d-block.
- Most of them form covalent compounds. Depending upon the number of valence electrons, the elements of the p-block exhibit variable oxidation states.
- These elements possess relatively higher ionization energy and the value tends to increase along the period but decrease down the group.
- Most of the elements show negative (except for some metals) as well as positive oxidation states (except for Fluorine). Besides group oxidation state, p block elements show a number of other oxidation states.
- In boron, carbon and nitrogen families, the group oxidation state is the most stable for the lighter elements in the group.
- One of the familiar characteristics of p-block elements is to show inert pair effect i.e. the tendency of being less availability for ns electron in bonding.
The inert pair effect increases down the group with an increase in the atomic number. The group oxidation state of group 13 elements is +3 but +1 oxidation state is almost constant for thallium. The group oxidation state for group 14 elements is +4 but +2 oxidation state is almost constant for the lead. A lower oxidation state which is 2 units less than the group oxidation state becomes gradually more constant for the heavier elements in each group.
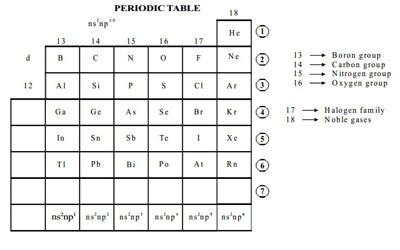
Elements in the p-block of the Periodic Table –
The elements in the p-block of the periodic table comprise a wide range of elements i.e. metals, non-metals, and metalloids. They are listed below –
Metals: Aluminium, Gallium, Indium, Thallium, Tin, Lead and Bismuth.
Non-Metals: Helium, Carbon, Nitrogen, Oxygen, Fluorine, Neon, Phosphorus, Sulphur, Chlorine, Argon, Selenium, Bromine, Krypton, Iodine, Xenon and Radon.
Metalloids: Boron, Silicon, Germanium, Arsenic, Antimony, Tellurium, Polonium and Astatine.