Applications of the Parachor
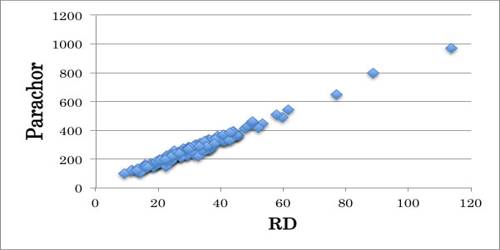
Parachor is the molecular weight of liquid times the fourth root of its surface tension, divided by the difference between the density of the liquid and the density of the vapor in equilibrium with it; essentially constant over wide ranges of temperature. It is an additive constitutive property of a molecule and is related to the molar volume and the surface tension. It is an empirical constant for a liquid that relates the surface tension to the molecular volume and that might be used for an assessment of molecular volumes under circumstances such that the liquids have the similar surface tension and for determinations of incomplete construction of compounds by adding values obtained for ingredient atoms and structural features.
The parachor has been used to determine the structure of a number of substances. An important example is p-benzoquinone for which the following two alternative structures were suggested:
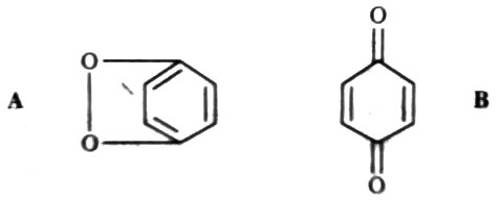
The parachor of structure A and B were calculated according to the data given in Table and found to be 230 and 206.9 respectively. The experimental value is 236.8, indicating that structure A is correct. A number of ether structural controversies have been settled with the help of parachor values.
It should be pointed out that anomalies in the parachor values have been noticed in a number of cases, viz., organometallic compounds. Since the advent of spectroscopic methods which give better results, the parachor is not used to any great extent in recent times.
The parachor of a biologically vigorous molecule is related to the aptitude of that molecule to permeate hydrophobic regions of cells, particularly cellular membranes. The parachor of a steroid can be calculated from its constituent atoms and bonds. An assessment of the parachor values of a large number of steroids shows that these values are associated with a number of dissimilar biological activities, from independent sources. The relative anti-inflammatory potencies of steroids by numerous analyze methods are directly proportional to their parachors.
Parachor method is applied to approximation surface tension of pure imidazolium-based ionic liquids at the dissimilar temperatures. For this prediction, a corresponding-states group-contribution method is proposed to estimate the surface tension of ionic liquids covering wide ranges of temperature and chain length.