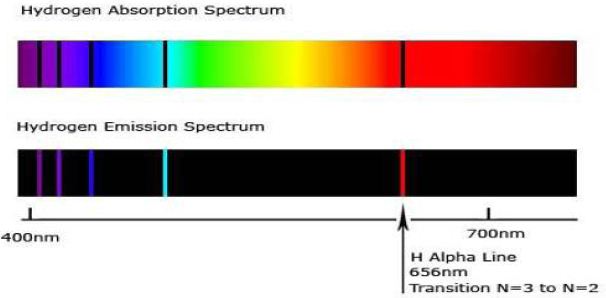
An element can also absorb light at specific wavelengths. An absorption spectrum can be obtained by passing a continuous radiation spectrum (white light) through a vapour of the gas. The absorption spectrum consists of a series of dark lines superimposed on the otherwise continuous spectrum (like the rainbow). The dark lines of the absorption spectrum coincide with the bright lines of the emission spectrum.
Absorption Spectra in Astronomy
Most stellar spectra are absorption spectra. Hot, high density objects give off a continuous spectrum. f this light then passes through a low density gas, photons of certain wavelengths can be absorbed — if the light is observed after it has passed through the gas, you will see an absorption spectrum. The continuous spectrum emitted by the Sun passes through the cooler gases of the Sun’s atmosphere:
– The various absorption lines can be used to identify elements in the solar atmosphere, and
– This led to the discovery of helium (named after Helios, god of the sun).
Uses of spectroscopy
By looking at a star or planet’s absorption spectrum we can learn about its composition. E.g. Sodium is only atom to produce 2 absorption lines in yellow part of the visible spectrum with wavelengths 589 and 589.6nm. The same is true for any chemical we heat up. Spectroscopy is an important technique for the quantitative analysis of many elements, particularly metals.












