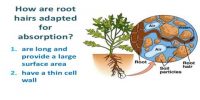
If a spoon of sugar dissolves in a glass of water, the sugar molecules spread throughout the whole glass of water; as a result the whole water becomes equally a sweet. If a spoon of salt is dissolved in a glass of water, it is seen that whole water becomes equally salted. Let us see what happens if some crystals of Potassium permanganate (KMnO4) is dropped into a glass of water. After some times it will be seen that the whole water in the glass becomes violet. Dropping a drop of ink in the glass may do this experiment.
Actually in a crystal of sugar, salt or potassium pennanganate there are any molecules compactly combined together. When the crystal is dropped in water the water molecules attract its molecules very strongly. As a result the crystal is completely dissolved in water and its molecules are spread throughout the water evenly. The molecules of sugar, salt or potassium pennanganate distributed throughout the water by the process diffusion.
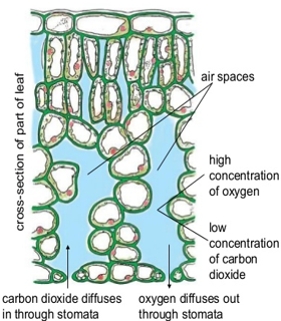
Diffusion is the net passive movement of particles (atoms, ions or molecules) from a region in which they are in higher concentration to regions of lower concentration. It continues until the concentration of substances is uniform throughout.
At a constant temperature and atmospheric pressure movements of the molecule of a substance from a region of high concentration to a region of low concentration is known as diffusion. The term diffusion means to distribute throughout.