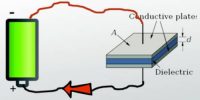
Fluorescence: When an atomic or molecular system is excited into higher energy state by absorption of energy, it returns back to lower energy state in a time less than 10-5 second and the system is found to glow brightly by emitting radiation of longer wavelength.
When ultra violet light is incident on certain substances, they emit visible light.
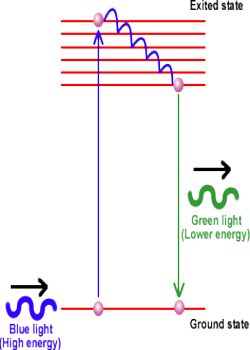
It may be noted that fluorescence exists as long as the fluorescing substance remain exposed to incident ultraviolet light and re-emission of light stops as soon as incident light is cut off.