On the basis of the experimental results of the Carnot’s engine scientists described how the heat is transferred from one system to the other. They have stated the nature of the transformation of heat energy in various ways, which refers the 2nd law of thermodynamics.
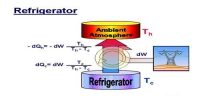
Statement of Clausius: It is not possible for a self acting machine to transfer heat energy from cold body to a heated body without the help of any external agency.
Statement of Plank: Construction of such machine is impossible which can convert all input heat energy to mechanical energy.
Statement of Lord Kelvin: It is not possible to continuous flow of heat from a body of lower temperature to a body of higher temperature.
Statement of Edser: The heat energy generally flows from a body of higher temperature to a body of lower temperature.