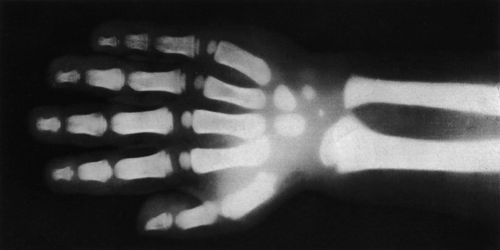
Properties of X-rays
X-rays or Röntgen rays are the revolutionary discovery of the nineteenth century. In 1985, famous German scientist Prof. W.K. Röntgen discovered these rays. We know that cathode rays are nothing but electrons having very high velocity. These high-velocity electrons when suddenly strike a solid metal. Their properties make them very useful in medicine. The detailed images they provide help to diagnose disease, while radiography uses high-energy X-rays to treat cancer.
The following properties of X-rays have been established from different experimental results:
(1) X-rays travel in straight line.
(2) X-rays are invisible. When ordinary light is incident on the retina, it creates the sensation of vision; but for X-rays this does not happen.
(3) It is electromagnetic transverse wave.
(4) Its wavelength is much smaller than the wavelength of ordinary light. Wavelength of ordinary light is of the order of 10-7 m or 1000 Å; but for X-rays wavelength is of the order of 10-10 m or., 1 Å.
(5) X-rays travel with uniform speed of 3 x 108 ms-1.
(6) Its penetrating power is extremely high.
(7) It has effect on the photographic plate.
(8) It creates fluorescence.
(9) These rays are not deflected by electric and magnetic fields.
(10) They ionize gas when they travel through the gas.
(11) X-rays show photoelectric effect. That means when X-rays are incident on metal surface electrons are emitted.
(12) Like ordinary light reflection, refraction interference, diffraction a polarization occur with X-rays.
(13) These rays can destiny living cells.
(14) The characteristics of genes are changed by their effect.
(15) Long exposure of the skin to X-rays is harmful which can destroy the white corpuscles of the blood.