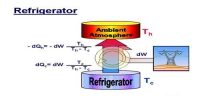
A thermodynamic cycle in which a working substance starting from a given initial thermal state undergoes two successive expansion, one isothermal and other is adiabatic and two successive compression, one is isothermal and the other is adiabatic and brought back to the initial state, is known as Carnot’s cycle.
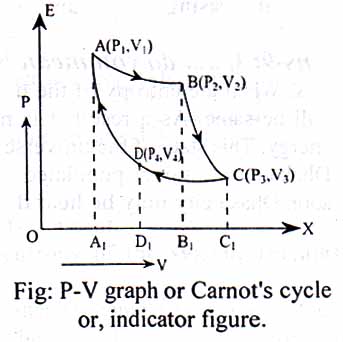
The graphical representation of carrot’s cycle is shown in the figure. The working steps are as follows:
1st Step: Isothermal expansion. In fig., AB is indicated as isothermal expansion. The work done for this step, W1 = area of AA1B1B.
2nd Step: Adiabatic expansion. In fig., it is shown by BC. The work done in this step, W2 = area of BCC1B1.
3rd Step: Isothermal compression. In fig, it is denoted by CD. The work done to this step, W3 = area of CC1D1D.
4th Step: Adiabatic compression. In fig., DA is the adiabatic compression. The work done in this step, W4 = area of DD1A1A
Therefore total work done by the engine is W.
So, W =W1 + W2 -W3 -W4 = area of ABCD.