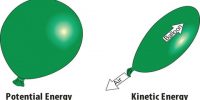
The temperature may be defined as the particular property which determines whether a system is in thermal equilibrium or not with its neighbouring system when they are brought into contact.
If we have a number of gaseous systems, whose different states are represented by their volumes and pressures V1, V2, V3 … and P1, P2, P3… etc., in thermal equilibrium with one another, we will have ϕ1 (P1, V1) = ϕ2 (P2, V2) = ϕ3 (P3, V3) and so on, where ϕ is a function of P and V. Hence, despite their different parameters of P and V, the numerical value of these functions or the temperature of these systems is same.