Adiabatic Compression of Carnot Cycle
The cylinder is now placed on the insulating stand and the piston is further moved down in such a way that the gas is compressed adiabatically to its initial volume V1 and pressure P1. As the gas is insulated from all sides heat produced raises the temperature of the gas to T1. This change is adiabatic and is represented by DA. Let W4 be the work done on the gas in compressing it adiabatically from a state D (V4, P4) to the initial state A (V1, P1).
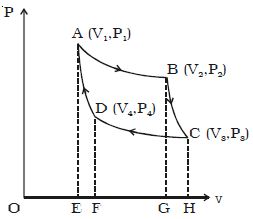
Then, W4 = v1∫v4 – PdV = -R/(γ-1).(T2-T1)
The negative sign indicates that work is done on the working substances.
So, W4 = R/(γ-1).(T1-T2) = Area DAEFD