The nucleus of Atom: The entire mass and positive charge are concentrated in a very small region at the centre of the atom known as the nucleus. The size of the nucleus is very small as compared to the size of the atom. The magnitude of positive charge on the nucleus is different for different atoms.
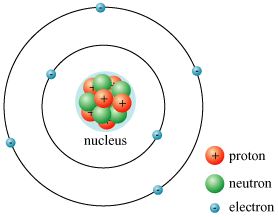
The nucleus, that intense inner core of the atom, surrounds both protons and neutrons. Electrons are exterior the nucleus in energy levels. Protons have a positive charge, neutrons have no charge, and electrons have a negative charge.