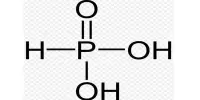
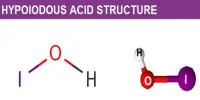
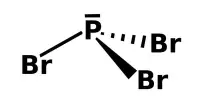
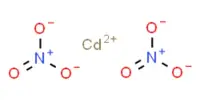
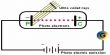
There is five process for the coagulation of a colloidal solution:
- Addition of electrolytic solution: When the electrolytic solution is added to a solution then the colloid’s particles attract the opposite changed electrolyte ions and neutralized. After neutralization colloid particles attract each other and precipitated. Example: When NaCl electrolyte is added As2S3 negative solution then yellow precipitate is formed. Na+ neutralize the negative colloid particles in this case.
- Mixing of two solutions having opposite charge: When two colloidal solution of opposite charged are mixed they neutralized to each other and precipitated forming large size particles. e.g. Fe(OH)3 and As2S3 solution mixing together are precipitated out.
- Vaporization: Lyophobic solutions are precipitated by vaporization. Because in high-temperature colloids particles are collided with each other more and more and form precipitation by closing together. e.g. Albumine is precipitated during heating in water.
- Electrophoresis: Applying the electric field to the colloid particles are neutralized and closing together from precipitation.
- Persistent dialysis: the Colloidal solution is stabilized due to the presence of few amount electrolytic solution. These electrolytic solutions are removed by persistent dialysis and remaining portion is precipitated out.