Hybridization is the process of formation of new equivalent orbitals from mixing u of two or more non equivalent atomic radius. The new orbitals are called hybrid orbitals.
Example:
Carbon atoms naturally have electron configuration 1s2 2s2 2p2.
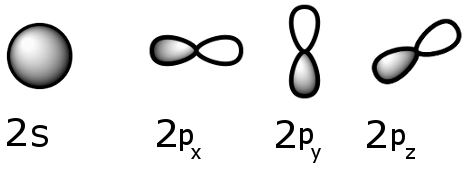
The four outermost electrons, i.e. those in the 2s and 2p sublevels are available to form chemical bonds with other atoms.
The 2s orbital is capable of holding up to two electrons, and there are three 2p orbitals, each capable of holding up to two electrons, which means the 2p orbitals can hold up to six electrons.