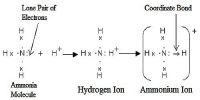
Chemical equilibrium: A chemical equilibrium in a system is the state of the system when there is no observable change in the concentration of the reactants and products. Concentration of the reactant and product as a function of time and attainment of equilibrium. At this state the concentrations of all species remain unchanged if other factors like temperature, pressure remain constant. The reaction can be started either from the reactant side or from the product side.
More Post
-

The New Humanoid Robot From Tesla Is About To Be Revealed – For Real This Time
-

Experimental Arrhenius Plot: Mathematical Example
-

Military Action in Radioactive Chernobyl Could Be Dangerous For People and the Environment
-
Pre-eclampsia can be Detected in Women with a Single Blood Sample
-

Bezos’s Blue Origin Loses Lunar Lander Appeal as US Government Upholds NASA SpaceX Contract
-

How University Students Perceive and Perform Kindness?
Latest Post
-

Nano-oscillator Achieves Record Quality Factor
-

Not Only Do Opposites Attract: A New Study Demonstrates That Like-Charged Particles Can Come Together
-

A Breakthrough in Single-photon Integration Shows Promise for Quantum Computing and Cryptography
-

Could the Sun be Conscious? Enter the Unorthodox World of Panpsychism
-

The Brains of Conspiracy Theorists Are Different: Here’s How
-

Baltimore’s Key Bridge Collapses Following Container Ship Collision