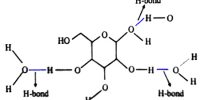
Catalyst: Catalyst is the substance which does not directly participate in the reaction but present in the reaction and increases or decrease the reaction rate. After complete the reaction catalysts remain unchanged
d-block elements work as catalyst.
d- block elements have the special electronic configuration. It has the empty space in the valance shell for that it donate or receive the electron and change their oxidation state. So it can participate electron exchanging reaction with other substance and create a alternative path a reaction which has less activation energy.
On the other hand p-block clement is non-metal, sub-metal and inert. These elements does not change the oxidation state. For that they participate the reaction and form compound.
d-block element work as catalyst. So it presents the reaction and increase the reaction rate an increase the profit of the industry.