The Crystal Systems
Crystalline solids exhibit different toms depending on the relative sizes of the ions or atoms, type of binding force, nature of stacking etc. any of the six or sometimes seven main groups into which crystals are commonly classified according to the relative lengths and inclinations of their axes or according to their respective symmetries.
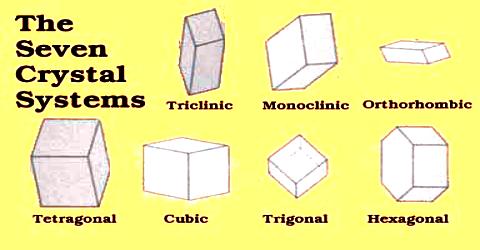
There are seven crystal systems that atoms can pack together to produce 3D space lattice. Each unit cell has characteristic parameters. A particular crystal may be formed from one of these unit cells and the seven different crystal structures thus formed are called crystal systems.
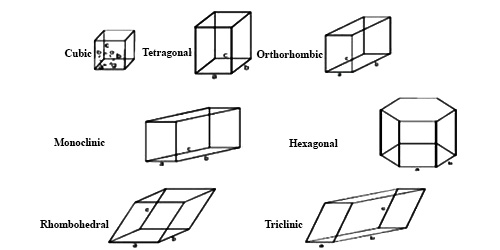
A characteristics feature of the crystals, large or small, of a given substances is that the angle between the corresponding faces of the crystals is the same. This is known as the “Law of the constancy of the Interfacial Angles”. The interfacial angles are the outer manifestation of the internal arrangement of the crystals. A study of the interfacial angles of different crystals is, therefore, of primary importance.
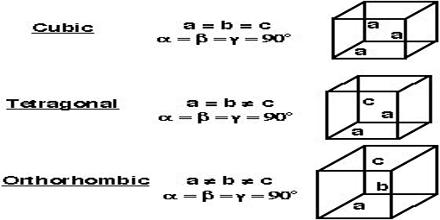
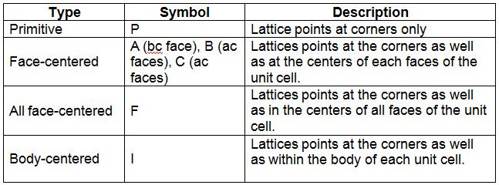
Description of different crystal systems: