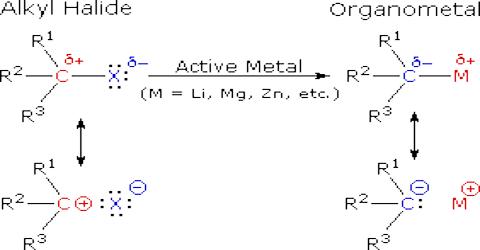
Reaction of Alkyl Halides with Metals:
(1) Reaction with sodium (Wurtz Reaction): Alkanes are produced by heating alkyl halides with metallic sodium in dry ether solution. The two alkyl groups join together to get a symmetrical alkane. Other metals have also been used to effect the Wurtz coupling, among them silver, zinc, iron, activated copper, indium and a mixture of manganese and copper chloride. This reaction is called Wurtz Reaction.
CH3 – CH2 – I + 2Na + CH3 – CH2 – I → Ether → CH3 – CH2 – CH2 – CH3 + 2NaI
The Wurtz reaction is limited to the synthesis of symmetric alkanes.
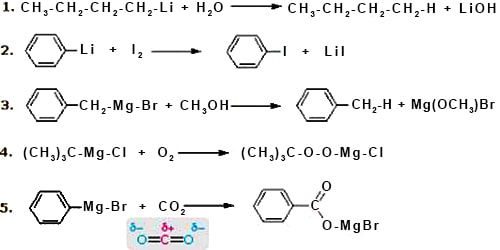
(2) Reaction with magnesium (Grignard Reagent): In dry ether, alkyl or aryl halides react with metallic magnesium to produce alkyl/aryl magnesium halides which is known as Grignard Reagent. A Grignard reagent has a formula RMgX where X is a halogen, and R is an alkyl or aryl (based on a benzene ring) group. For the purposes of this page, we shall take R to be an alkyl group.
CH3I + Mg →Ether→ CH3MgI
From Grignard reagent we get: Hydrocarbon; Alcohol and Carboxylic acid.