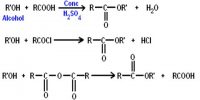
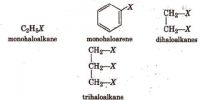
In 1927 Davisson and Germer observed that, a beam of electrons obtained from a heated tungsten filament is accelerated by using a high positive potential. When this fine beam of accelerated electron is allowed to fall on a large single crystal of nickel, the electrons are scattered from the crystal in different directions. The diffraction pattern so obtained is similar to the diffraction pattern obtained by Bragg’s experiment on diffraction of X-rays from a target in the same way (Figure).
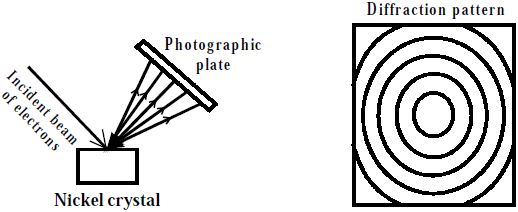
Fig: Electron diffraction experiment by Davisson and Germer
Since X-rays have wave character, therefore, the electrons must also have wave character associated with them. Moreover, the wave length of the electrons as determined by the diffraction experiments were found to be in agreement with the values calculated from de-Broglie equation. From the above discussion, it is clear that an electron behaves as a wave.