Identification of Copper (Cu):
Copper is a changeover metal, one of numerous elements found in rows 4 through 7 between Groups 2 and 13 in the periodic table. The periodic table is a chart that shows how chemical elements are related to each other.
Wet test:
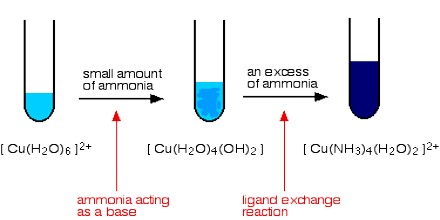
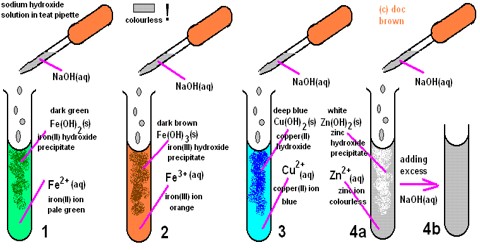
i) NH4+ solution is added slowly. Deep blue solution of [Cu(NH3)4] SO4 is obtained.
CuSO4 + NH4OH → CuSO4.Cu(OH)2 ↓ (Light blue) + (NH4)2 SO4
ii) KI is added, white ppt. of Cu2I2 is obtained. The color of the solution is brown due to presence of free I2.
2CuSO4 +4KI → Cu2I2 ↓ (White) + 2K2 SO4 + I2
iii) If Potassium Ferro cyanide solution is added in copper salts, a reddish brown ppt. is obtained.
2CuSO4 + K4 [Fe(CN)6] → Cu2 [Fe(CN)6] ↓ (Reddish brown ppt) + 2K2SO4
Copper is a reasonably dynamic metal. It dissolves in most adds and in alkalis. An alkali is a chemical with properties opposite those of an acid. Sodium hydroxide, generally found in bleach and drain cleaners like Drano, is an example of an alkali. An vital chemical property of copper is the way it reacts with oxygen. In moist air, it combines with water and carbon dioxide. The product of this reaction is called hydrated copper carbonate