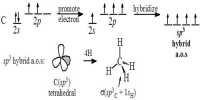
Baeyer’s Solution Test: The given sample is dissolved in alcohol. A few drops of aqueous (neutral/ alkaline) solution of KMnO4 (1 % solution) added to the mixture. A rapid decolonization of the pink color of the solution confirms the presence of unsaturated compound or double bond. It is also known as Hydroxylation reaction.

Example: When ethylene gas is shaken with alkaline solution of potassium permanganate, rapid decolourisation of the pink color of the solution takes place due to the formation of propylene glycol, which confirms the presence of ethylene. This is known as Baever’s test.