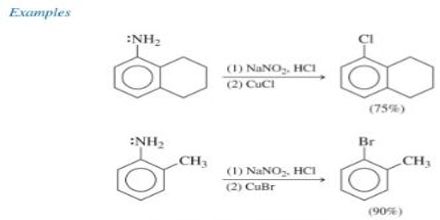
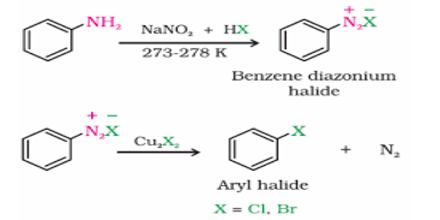
Sandmayer Reaction: If the aqueous solution of diazonium salt is heated at 100 C with cuprous salt and its similar hydracids, the diazo group is substituted by the acidic group of cuprous salt to produce benzene derivatives. This chemical reaction influenced by cuprous ion is known as Sandmeyer reaction. This is a reaction for preparing aromatic halides or cyanides from a salt with a cuprous halide. It is named after the Swiss chemist Traugott Sandmeyer.
For example, if benzene diazonium chloride is heated at 100°C with CuBr and HBr; bromo benzene is produced.
If phenyl cyanide produced in the Sandmayer Reaction is hydrolyzed- benzoic can be obtained.