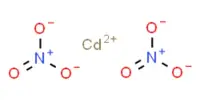
Boiling point of Ammonia (NH3) is more than Phosphine (PH3):
The size of nitrogen atom is less than phosphorus atoms. So, nitrogen is more electronegative element. That’s why the electrons of N – H bond come closer to N-atom than the electrons of P-H bond that come closer to P-atom. So, the polarity of N-H bond of NH3 is more than that of P-H bond of PH3. This causes the tendency of formation of hydrogen bond in NH3 more than that in PH3. As NH3 molecules are joined by hydrogen bond, so more energy is needed to break down N-H bond of NH3 than P-H bond of PH3.
So, the boiling point of NH3(- 33°C) is higher than that of PH3(- 87°C).