Emission and absorption spectra are evidence that the electrons within an atom have quantised energies i.e. an electron cannot possess a continuous range of values for energy but must have specific values called energy levels. The lowest energy level is called the ground state (E.). Any higher energy level is called an excited state.
Bohr model
In 1913 Niels Bohr (1885-1962) developed a theory to explain light emission and absorption. The Bohr model of the atom pictures electrons circling the nucleus at a fixed radius – in an energy shell. Bohr’s theory works well for the one-electron hydrogen atom, but note that more sophisticated theories are needed to explain multi-electron atoms.
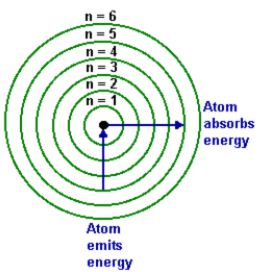
The energy shells are labelled with a principal quantum number; n=1, n=2 etc. The closer the electron’s orbit is to the nucleus, the stronger the attractive force. The lowest energy electrons are those nearest the nucleus in the n = 1 shell. Extra energy would need to be added to enable them to pull away from the nuclear attraction into an excited state.











