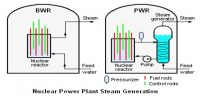
Binding energy of Nucleus
Whenever a nucleus is formed, certain mass is converted into energy. Hence for atom, the atomic mass is lower than the sum of masses of protons, neutrons and electrons present. The difference in mass is termed as “mass defect”. This is the measure of the binding energy of proton and neutron in the nucleus. The relationship between mass – energy is explained by Einstein equation ΔE = Δm C2.